Hair Transplant and Restoration for Las Vegas, Henderson, & Nearby Areas in Nevada
In order to obtain maximum density now and in the future, it is important to use adjunctive non-surgical therapy to preserve existing native hair. Non-surgical hair replacement or hair growth therapy may bring back some lost hair, but just as important, it may slow down or stop future loss.
The use of non-surgical therapy can affect surgical planning in a number of ways. Sometimes the response is so good that patients decide they don’t need surgery. Sometimes it improves the situation so a smaller, less aggressive procedure is needed. Even if non surgical therapy is only minimally effective, a small amount of regrowth can significantly improve the appearance of density in transplanted areas.
Minoxidil Hair Growth Treatment
Minoxidil (Rogaine®) is a topical application approved for both men and women. It has been shown to either reduce hair loss or promote hair growth in about 80% of patients. Minoxidil acts as a direct anagen-prolonging agent. The original product, however, was oily and messy, and the propylene glycol additive could cause irritation in some patients. Recently, a new 5% foam delivery system has been released without any propylene glycol. It is much easier to use and better accepted by patients. Although the 5% is labeled for men, it can be used by both men and women.
The active ingredient in Rogaine, minoxidil, was initially used to treat high blood pressure in the drug Loniten. A side effect of the drug was the reversal or deceleration of hair loss. In 1988, the Upjohn Corporation received approval to distribute Rogaine for men. In 1991, the Food and Drug Administration approved a Rogaine formula for women. Rogaine was only available through prescription until 1996, when Rogaine was approved for sale over the counter.
Application
Rogaine should be applied as directed by a physician or as stated on the packaging. The scalp must be clean and dry prior to application. Rogaine comes with a dropper for application of the liquid product. A typical treatment is 1 ml, which is inserted into the dropper and applied directly to the scalp two times daily. Rogaine’s foam formula can be applied directly to the scalp with the fingers. Rogaine does not need to be washed out after application.
Side Effects
Most side effects of Rogaine occur on the scalp and are mild. The treatment can irritate, burn or sting the scalp and cause itchiness. The alcohol content may also cause dryness in the scalp that can result in dandruff. The foam preperation does not irrate the scalp as much as the alcohol based product, but is more expensive. The drug may also cause an initial reaction of increased hair loss. More serious side effects of Rogaine are rare but include a rapid heartbeat, dizziness and swelling of the hands, feet or face. An allergic reaction to Rogaine can result in a rash and trouble breathing.
Warning
Rogaine is not recommended for pregnant women. Pregnant women should discuss taking this drug with their physicians. It is also unknown if the active ingredients in Rogaine can pass through a woman’s body into breast milk. If the scalp is irritated or has open wounds, Rogaine should not be used. This drug is also not recommended for use on any parts of the body aside from the scalp. People with heart, kidney or liver disease should seek medical advice before taking Rogaine.
Propecia (Finasteride) Hair Growth Treatment
Propecia® (Finasteride) is a drug used to treat male pattern hair loss. Finasteride was initially approved by the FDA in 1992 under the brand name Proscar, for the treatment of an enlarged prostate. In 1997, the FDA approved finasteride for the treatment of male pattern baldness (MPB) under the brand name Propecia. This drug treats male pattern hair loss by blocking the body’s production of a male hormone called DHT. Finasteride is effective only for as long as it is taken; the hair gained or maintained is lost within 6–12 months of ceasing therapy. In clinical studies, finasteride, was shown to work on both the crown area and the hairline, but is most successful in the crown area. Finasteride is not indicated in women and is contraindicated in women who are potentially able to have children due to birth defects.
Hair loss for both men and women is usually due to some hormonal changes in the body. DHT (Dihydrotestosterone) is the natural metabolite of the human body mainly responsible for hair loss. DHT exists when the male hormone Androgen gets involved with an enzyme called the 5 alpha reductase and combines with the male hormone testosterone. When DHT is abundant in an individual, it reaches the hair follicles and tends to prevent proteins, vitamins and minerals from providing the needed nourishment to sustain life in the hair of these follicles. The hair follicles then tend to reproduce at a slower rate with shortened growing phases and longer resting stages until eventually they stop growing at all. Because DHT is responsible for about 95% of hair loss, men or women who are genetically pre-disposed in producing more DHT than others are at higher risk of MPB.
Learn more about how Dr. Brown and Hair Transplant Vegas can help you restore lost hair by calling our Henderson, Nevada, office at 702-990-1975 , or contacting us online.
Hair Transplant Vegas Is proud to announce the arrival of the LaserCap, and Dr. Brown is the only physician in Las Vegas and Nevada to offer this exciting device. Through low light laser therapy, we are able to stimulate cell growth and blood flow in and around the follicles, improving hair regeneration. For the full science of photobiology and low level laser therapy, please follow this link: https://www.photobiology.info/Hamblin.html.
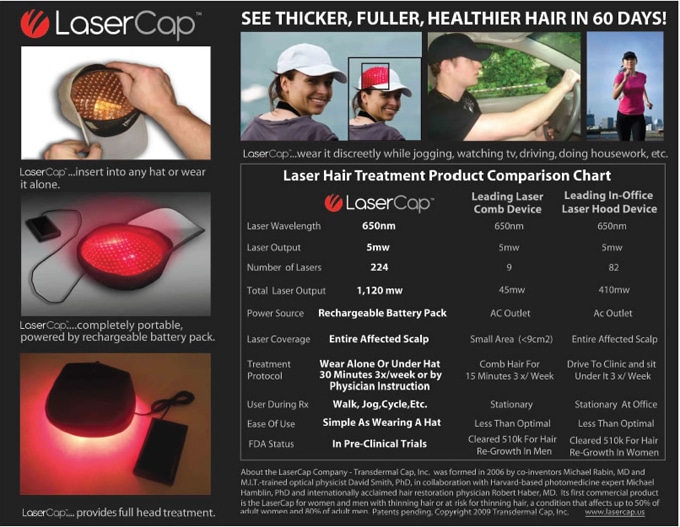
Find out if LaserCap treatment is right for you. Contact Dr. Steve Brown and his team at Hair Transplant Vegas online or by phone at 702-990-1975 to arrange a consultation.

